The menopause transition is a vulnerable cardiometabolic period in a woman’s life. Characterized by unfavorable changes in blood pressure, cholesterol, and metabolic function, many women simultaneously endure a host of symptoms including but not limited to sleep disruption, hot flashes, night sweats, mood changes, and weight gain. These symptoms impair quality of life and functional capacity. Menopausal hormone therapy (MHT) is the most effective treatment for moderate to severe vasomotor (VSM) and genitourinary (GU) symptoms but can be associated with increased cardiovascular (CV) and thromboembolic risk in high-risk patients.
Unfortunately, the potential risks of MHT have led to clinician and patient hesitancy in the use of MHT for appropriate patients, with many women ultimately suffering through uncomfortable VSM and GU symptoms. By reviewing the evidence of MHT and summarizing the current multi-society guidelines, we hope to provide a framework for risk assessment which can be used to select patients who may be acceptable candidates for MHT, while highlighting those who may benefit from CV risk reduction. While menopause is a time of heightened CV risk, it poses an opportunity for implementing risk reduction and prevention therapies. The concern about the risk of CV events MHT comes from the Women’s Health Initiative (WHI) published in the early 2000’s, which was a large research program aimed at studying major causes of morbidity and mortality in postmenopausal women. It was a large, randomized trial evaluating the safety of MHT in healthy postmenopausal women. This study demonstrated increased risk in the hormone therapy group, which was assigned to estrogen plus progestin, compared to placebo. Specifically, women received conjugated equine estrogen (CEE) 0.625 mg/day and medroxyprogesterone acetate (MPA) 2.5 mg/day versus placebo and were followed for safety and efficacy outcomes including coronary disease and breast cancer. The trial was stopped early due to excess in breast cancer in the treatment group. Additionally, stroke and pulmonary embolism were more common in the hormone group. Given these findings, MHT recommendations and prescribing patterns have decreased significantly in the years since this publication. However, since these findings, multiple retrospective studies evaluating subgroups of the WHI data have revealed nuance regarding the safety of MHT, based on the time since menopause onset and starting MHT therapy.
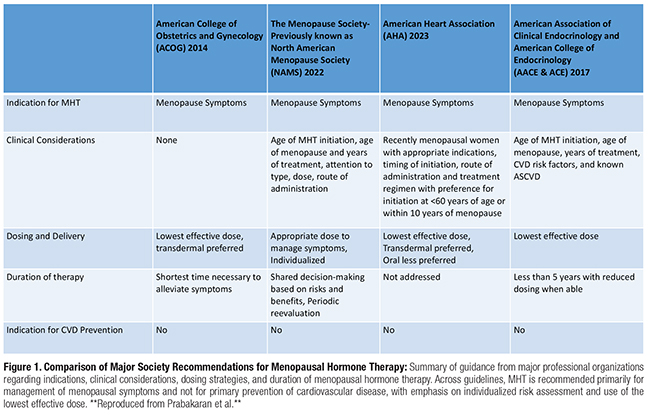 The “Timing Hypothesis” is a concept that was developed from these retrospective analyses and is based on the observation that MHT is associated with more risk when initiated after a significant amount of time since menopause onset. In women younger than 60 years or within 10 years of menopause, MHT was associated with lower CV risk. The underlying biologic mechanism proposal suggests that in women with minimal atherosclerosis, estrogen has beneficial effects on healthy endothelial function, lipid metabolism, and inflammation. In contrast, initiating MHT in older women or those with established subclinical/clinical atherosclerosis contributes to increased cardiovascular events. The timing hypothesis serves as an explanation to the WHI findings of harmful effects in older participants with null to beneficial effects in younger participants. This theory has been partially supported by the Early versus Late intervention Trial with Estradiol (ELITE) which evaluated carotid intima-mediated thickness (CIMT), a surrogate for CV disease, in those women randomized to early MHT (<6 years since menopause onset) or late MHT (>10 years since menopause onset). After 5 years of follow up, the women in the early group had less progression of CIMT compared to placebo, whereas those in the late group had no difference in progression compared to placebo.
The “Timing Hypothesis” is a concept that was developed from these retrospective analyses and is based on the observation that MHT is associated with more risk when initiated after a significant amount of time since menopause onset. In women younger than 60 years or within 10 years of menopause, MHT was associated with lower CV risk. The underlying biologic mechanism proposal suggests that in women with minimal atherosclerosis, estrogen has beneficial effects on healthy endothelial function, lipid metabolism, and inflammation. In contrast, initiating MHT in older women or those with established subclinical/clinical atherosclerosis contributes to increased cardiovascular events. The timing hypothesis serves as an explanation to the WHI findings of harmful effects in older participants with null to beneficial effects in younger participants. This theory has been partially supported by the Early versus Late intervention Trial with Estradiol (ELITE) which evaluated carotid intima-mediated thickness (CIMT), a surrogate for CV disease, in those women randomized to early MHT (<6 years since menopause onset) or late MHT (>10 years since menopause onset). After 5 years of follow up, the women in the early group had less progression of CIMT compared to placebo, whereas those in the late group had no difference in progression compared to placebo.
This emphasizes the importance of understanding the distinction between absolute vs. relative risk. While the relative increased risk of an event like a stroke may have been statistically significant in early analyses, the absolute risk in a healthy recently menopausal woman remained low. From the provider standpoint, this is central to understanding who is safe to receive MHT
 The menopausal transition is often discussed in the context of symptoms, but it is also a period of increased cardiometabolic risk. This period is characterized by a rise in LDL cholesterol, an increase in central adiposity, a steepening of blood pressure trajectories, and an uptick in insulin resistance. Therefore, clinic visits should be used strategically to identify and target cardiovascular risk factors
The menopausal transition is often discussed in the context of symptoms, but it is also a period of increased cardiometabolic risk. This period is characterized by a rise in LDL cholesterol, an increase in central adiposity, a steepening of blood pressure trajectories, and an uptick in insulin resistance. Therefore, clinic visits should be used strategically to identify and target cardiovascular risk factors
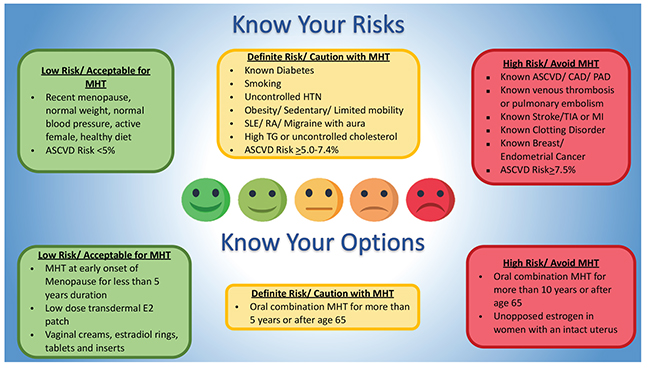
A comprehensive CV risk assessment should be performed that includes calculating an ASCVD risk score or the new Predicting Risk of Cardiovascular Disease EVENTs (PREVENT) equation and taking a detailed reproductive history. A history of adverse pregnancy outcomes (hypertensive disorders of pregnancy, eclampsia, preeclampsia, gestational diabetes) or early menopause confers a significantly higher lifetime risk of ischemic heart disease. These “risk-enhancers” can reclassify women from borderline to intermediate risk and thus prompt more aggressive prevention strategies. The risk assessment should inform how MHT can be considered as a part of a broader cardiometabolic strategy for appropriately selected women, but MHT is not recommended for the sole purpose of preventing coronary heart disease in women of any age.
So what are the guidelines saying? Luckily, the consensus has moved away from a “one-size-fits-all” warning that has been the dominant narrative. The American College of Obstetricians and Gynecologist (ACOG), the Menopause Society, and the Endocrine Society recent position statements were in agreement. They outline the appropriate candidate for MHT:
1. Are younger than 60 years old OR within 10 years of menopause onset.
2. Have moderate-to-severe vasomotor or genitourinary symptoms
3. Have low-to-intermediate ASCVD risk
4. Have no personal history of breast cancer, estrogen-sensitive malignancy, venous thromboembolism, or active liver disease
Once the appropriate candidate for MHT is identified, the next consideration is what formula and route is appropriate. MHT administration route modulates risk, particularly for thrombotic events. Oral estrogens undergo metabolism in the liver, which can increase the production of clotting factors and triglycerides. Transdermal estradiol (patches, gels) bypasses the liver allowing for a more neutral effect on lipids and coagulation.
The transdermal route is often preferred for those with risk factors of obesity, migraine with aura, or smoking due to lower VTE risks. Low-dose vaginal estrogen therapy (creams, rings, tablets) is an effective option due to their minimal systemic absorption and negligible cardiovascular risk in those with primarily genitourinary symptoms.
A simplified, stepwise approach may assist clinicians:
1. Risk Stratify – Calculate the CV risk and screen for a history of VTE, migraine with aura, cancer, and liver disease.
2. Assess Timing – Determine age and years since menopause.
3. Determine Goals: relief from moderate-to-severe vasomotor, genitourinary symptoms, or both
4. Choose route and dose: Aim for low-dose transdemal estradiol for systemic therapy in women with CV risk factors. The goal is to find the lowest effective dose.
5. Engage in shared decision-making: Discuss individualized risk and benefits. Reassess periodically and use the shortest duration consistent with symptom control.
 Considerable progress has been made in understanding CV risk with MHT when used appropriately, but important gaps in our knowledge remain. Further investigation is needed on the risk in women with elevated CV risk and who require long-term therapy beyond age 60. CV Risk calculators are limited and still often underestimate younger women and overestimate CV risk in older women. Significant health disparities persist in research participation and access to care for women of color who bare disproportion burden of cardiometabolic disease.
Considerable progress has been made in understanding CV risk with MHT when used appropriately, but important gaps in our knowledge remain. Further investigation is needed on the risk in women with elevated CV risk and who require long-term therapy beyond age 60. CV Risk calculators are limited and still often underestimate younger women and overestimate CV risk in older women. Significant health disparities persist in research participation and access to care for women of color who bare disproportion burden of cardiometabolic disease.
As physicians and health care providers, effective communication and patient education is needed to address the historical concerns regarding MHT safety and efficacy. Communicating that MHT is prescribed primarily for menopause symptom relief and is not indicated for CV benefit. The discussion should focus on individualized risks, treatment goals, and routine follow up to ensure the shortest duration and lowest dose to achieve symptoms relief and long-term safety.
MHT and cardiovascular risk is a powerful example of how science and medicine can evolve over time. What was once interpreted as universal harm has evolved into a more nuanced understanding of age, timing, formulation, and individual risk as key to achieving more optimal patient care. As millions navigate menopause each year, this more contemporary approach is paramount to supporting such an important transition in one’s life. Through understanding menopause as the ideal opportunity for CV risk prevention and establishing interdisciplinary collaboration between cardiologists, primary care physicians, endocrinologists and gynecologists, we can ensure that our patients receive comprehensive, compassionate, and evidence-based care during this pivotal stage of life.
References
Prabakaran S, Schwartz A, Lundberg G. Cardiovascular risk in menopausal women and our evolving understanding of menopausal hormone therapy: risks, benefits, and current guidelines for use. Ther Adv Endocrinol Metab. 2021 Apr 30;12:20420188211013917. doi: 10.1177/20420188211013917. PMID: 34104397; PMCID: PMC8111523.
Schwartz AM, Sweeney ME, Dolan M, Lundberg GP. Menopause Hormone Therapy: 30 Years of Advances and Controversies. Endocr Pract. 2025 Dec;31(12):1642-1648. doi: 10.1016/j.eprac.2025.10.011. Epub 2025 Oct 26. PMID: 41151698.
American College of Obstetricians and Gynecologists. Practice Bulletin No. 141: Management of Menopausal Symptoms. Obstet Gynecol. 2014;123(1):202–216. doi:10.1097/01.AOG.0000441353.20693.78
The Menopause Society (formerly NAMS) 2022 Hormone Therapy Position Statement Advisory Panel. The 2022 hormone therapy position statement of The Menopause Society. Menopause. 2022;29(7):767–794. doi:10.1097/GME.0000000000002028
Cho L, Kaunitz AM, Faubion SS, et al. Rethinking menopausal hormone therapy: for whom, what, when, and how long? Circulation. 2023;147(7):597–610. doi:10.1161/CIRCULATIONAHA.122.061559
Cobin RH, Goodman NF; AACE Reproductive Endocrinology Scientific Committee. American Association of Clinical Endocrinologists and American College of Endocrinology position statement on menopause—2017 update. Endocr Pract. 2017;23(7):869–880. doi:10.4158/EP171828.PS


