Ventricular tachycardia (VT) remains a leading cause of morbidity and mortality in patients with structural heart disease. Implantable cardioverter-defibrillators (ICDs) reduce sudden cardiac death but do not prevent recurrent VT, which leads to hospitalizations and impaired quality of life. Catheter ablation has emerged as a central strategy in VT management, with multiple randomized controlled trials shaping its role over the past two decades. While radiofrequency (RF) ablation remains the standard of care, limitations in lesion depth and substrate complexity within inaccessible regions have driven the development of novel strategies and adjunctive technologies to overcome these challenges. This review summarizes recent VT ablation trials, highlights contemporary challenges with RF ablation, and explores novel technologies on the horizon.
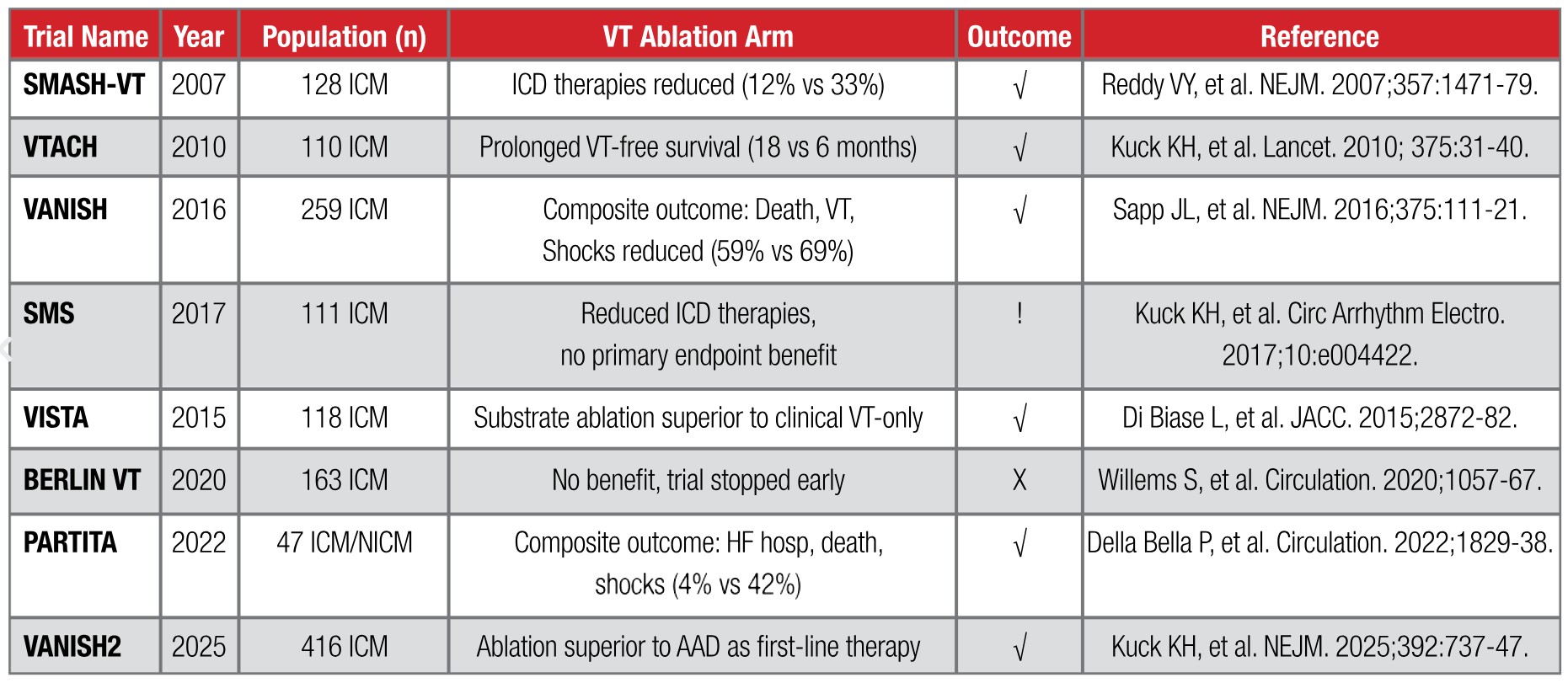
Ventricular tachycardia is frequently encountered in patients with ischemic cardiomyopathy (ICM) and non-ischemic cardiomyopathy (NICM) with scar, contributing to recurrent hospitalizations, ICD shocks, and worsening heart failure (HF) outcomes.1 While antiarrhythmic drug (AAD) therapy provides partial benefit, long-term use is limited by toxicity and incomplete efficacy.2 Catheter ablation has emerged as a cornerstone of therapy; however, its procedural complexity, modest success rates, and recurrence risk underscore the need for innovation. Table 1 lists several important trials focusing on catheter ablation of VT.
Table 1: Trials in VT ablation
Limitations of RF Ablation
Conventional RF ablation creates lesions with a mean depth of ~5 mm after resolution of surrounding edema, often insufficient for transmural or intramural scar circuits.10 Challenges include inaccessible regions (LV summit, papillary muscles, intramural scar/foci), and collateral risk (coronary arteries, phrenic nerve). Adjunctive strategies include half-normal saline irrigation, bipolar RF ablation, alcohol ablation, and needle-based RF delivery. These techniques have been utilized with variable success.
Novel and Emerging Technologies
Pulsed Field Ablation (PFA) is an emerging non-thermal ablation technology that uses high-voltage, ultra-short electrical pulses to induce irreversible electroporation of myocardial cell membranes, resulting in selective cell death. Unlike radiofrequency or cryo ablation, PFA primarily affects myocardial tissue while sparing adjacent non-cardiac structures such as the phrenic nerve, esophagus, and coronary arteries, which is particularly advantageous in the ventricular myocardium. Early preclinical and first-in-human studies suggest that PFA may enable rapid lesion creation, improved safety, and durable conduction block in VT ablation. Ongoing clinical trials are evaluating its efficacy, safety, and long-term outcomes compared with conventional energy sources.
Stereotactic Body Radiotherapy (SBRT), also referred to as stereotactic arrhythmia radioablation (STAR), is a novel noninvasive technique under investigation for treatment of refractory VT. SBRT delivers highly focused, high-dose ionizing radiation to arrhythmogenic myocardial scar regions identified through electroanatomic mapping and advanced imaging, creating targeted fibrosis and electrical isolation. This approach avoids the procedural risks of catheter ablation and may be an option for patients who are poor candidates for invasive therapy. Early clinical studies and case series, such as the ENCORE-VT trial, have demonstrated significant reductions in VT burden and ICD shocks, though concerns remain regarding long-term safety, delayed radiation effects, and optimal patient selection. Ongoing prospective trials aim to establish efficacy, refine dosing protocols, and evaluate late toxicities.
Cardiac sympathetic denervation (CSD), or surgical sympathectomy, is an important adjunctive therapy for patients with refractory VT or electrical storm despite antiarrhythmic drugs and catheter ablation. By interrupting sympathetic input to the heart—typically via left or bilateral stellate ganglion and thoracic sympathetic chain ganglion resection—CSD reduces adrenergic drive, thereby decreasing arrhythmia triggers. Clinical studies and case series have shown that CSD can significantly reduce VT burden, ICD shocks, and arrhythmia-related hospitalizations, particularly in patients with structural heart disease and scar-mediated VT. While not curative, sympathectomy offers a palliative option and ongoing investigations are exploring neuromodulation strategies.
Ventricular Intramyocardial Navigation for Tachycardia Ablation Guided by Electrograms (VINTAGE) is a novel invasive technique designed to target deep intramural substrate. The method involves steering a guidewire/microcatheter assembly through the ventricular myocardium, accessed typically from the right ventricle, with trajectory guided by electrogram recordings, fluoroscopy, and electroanatomic mapping. Once positioned in the target intramyocardial site, radiofrequency energy is used to produce deep lesions. An initial human experience in patients with prior failed endocardial ablations demonstrated high technical success, good arrhythmia reduction (VT/PVC burden), with acceptable safety profile.
Catheter ablation has evolved from a salvage intervention to a central therapeutic modality for VT, supported by robust clinical trial evidence. Early intervention, extensive substrate modification, and integration of novel technologies are redefining the therapeutic landscape. While challenges remain, ongoing clinical trials promise to transform VT management over the next decade.
References
1. Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation 2018;138(13), e272-e391.
2. Sapp JL, Tang ASL, Parkash R, Stevenson WG, Healey JS, Gula LJ, Nair GM, the VANISH2 Study Team. Catheter ablation or antiarrhythmic drugs for ventricular tachycardia. N Engl J Med 2025;392(13), 737–747.
3. Reddy VY, Reynolds MR, Neuzil P, et al. Prophylactic catheter ablation for the prevention of defibrillator therapy. N Engl J Med 2007;357(26):2657-2665.
4. Kuck KH, Schaumann A, Eckardt L, et al. Catheter ablation of stable ventricular tachycardia before defibrillator implantation in patients with coronary heart disease (VTACH): a multicentre randomised controlled trial. Lancet 2010;375(9708):31-40.
5. Sapp JL, Wells GA, Parkash R, Stevenson WG, et al. Ventricular tachycardia ablation versus escalation of antiarrhythmic: The VANISH trial. N Engl J Med 2016;375(2):111-121.
6. Kuck KH, Tilz RR, Deneke T, et al. Impact of substrate modification by catheter ablation on implantable cardioverter-defibrillator interventions in patients with unstable ventricular arrhythmias and coronary artery disease: results from the multicenter randomized controlled SMS (Substrate Modification Study). Circ Arrhythm Electrophysiol 2017;10(3):e004422.
7. Di Biase L, Burkhardt JD, Lakkireddy D, et al. Ablation of stable ventricular tachycardia versus substrate ablation in ischemic cardiomyopathy: The VISTA randomized multicenter trial. J Am Coll Cardiol 2015;66(25):2872-2882.
8. Willems S, Tilz RR, Stevens D, et al. Preventive or deferred ablation of ventricular tachycardia in patients with ischemic cardiomyopathy and implantable defibrillator (BERLIN VT): A multicenter randomized trial. Circulation 2020;141(13):1057-1067.
9. Della Bella P, Baratto F, Vergara P, et al. Does timing of ventricular tachycardia ablation affect prognosis in patients with an implantable cardioverter-defibrillator? Results from the PARTITA trial. Circulation 2022;145:1829-1838.
10. Glashan CA, Stevenson WG Zeppenfeld Z. Lesion size and lesion maturation after radiofrequency catheter ablation in patients with nonischemic cardiomyopathy. Circ Arrhythm Electrophysiol 2021;14(8):e009808.
11. Bennett R, Campbell T, Byth K, Turnbull S, Kumar S. Catheter ablation using half-normal saline and dextrose irrigation in an ovine ventricular model. JACC Clin Electrophysiol 2021;7(10):1229–1239.
12. Ravi V, Winterfield J, Liang JJ, et al. Solving the reach problem: A review of present and future approaches for addressing ventricular arrhythmias arising from deep substrate. Arrhythm Electrophysiol Rev 2023;12:e04.
13. Brugada P, de Swart H, Smeets JL, Wellens HJ. Transcoronary chemical ablation of ventricular tachycardia. Circulation 1989;79(3):475–482.
14. Zipes DP, Waller BF, Jalife J. Intracoronary ethyl alcohol or phenol injection ablates aconitine-induced ventricular tachycardia in dogs. J Am Coll Cardiol 1987;10:1342–1349.
15. Sapp JL, Beeckler C, Pike R, Parkash R, Gray C, Stevenson WG, et al. Initial human feasibility of infusion needle catheter ablation for refractory VT. Circulation 2013;128(21):2289–2295.
16. Packer DL, Wilber DJ, Kapa S, Dyrda K, et al. Ablation of refractory VT using intramyocardial needle delivered heated saline-enhanced radiofrequency energy: a first-in-man feasibility trial. Circ Arrhythm & Electrophysiol 2022;15:e10347.
17. Martin CA, Zaw MT, Jackson N, Morris D, et al. First worldwide use of pulsed-field ablation for ventricular tachycardia ablation via a retrograde approach. Journal of Cardiovasc Electrophysiol 2023;1-4.
18. Lozano-Granero C, Reddy VY, et al. Case series of ventricular tachycardia ablation with a single-shot pulsed field ablation pentaspline catheter. JACC: Clinical Electrophysiol 2023;9:1990-4.
19. Zhang Z, et al. Pulsed field ablation: a promising approach for ventricular tachycardia — review of preclinical evidence and early clinical experience. Int J Cardiol 2024;407:131985.
20. Cuculich PS, Schill MR, Kashani R, Mutic S, Robinson CG, et al. Noninvasive cardiac radiation for ablation of ventricular tachycardia. N Engl J Med 2017;377(24):2325-2336.
21. Lloyd MS, Wight J, Schneider F, et al. Clinical experience of stereotactic body radiotherapy for refractory ventricular tachycardia. JACC Clin Electrophysiol 2020;6(8):958-969.
22. Robinson CG, Samson PP, Moore KMS, Cuculich PS, et al. Phase I/II trial of electrophysiology-guided noninvasive cardiac radioablation for ventricular tachycardia. Circulation 2019;139(3):313-321.
23. Vaseghi M, Barwad P, Malavassi Corrales FJ, et al. Cardiac sympathetic denervation in patients with refractory ventricular arrhythmias or electrical storm: intermediate and long-term follow-up. Heart Rhythm 2014;11(3):360-366.
24. Hanna DB, Karimianpour A, Mamprejew N, Fiechter C, Sharma D, et al. The role of cardiac sympathetic denervation for ventricular arrhythmias: an updated systematic review and meta-analysis. J Interv Card Electrophysiol 2025;68(2):415-425.
25. Bhatia NK, Halaby RN, Bruce CG, Shah AD, Badhwar N, Greenbaum AB, Babaliaros VC, Lederman RJ, et al. Ventricular intramyocardial navigation for tachycardia ablation guided by electrograms (VINTAGE): Initial Human Experience. JACC: Clin Electrophysiol 2025;S2405-500X.
 Ahmadreza Karimianpour, DO, FACC, FHRS
Ahmadreza Karimianpour, DO, FACC, FHRS
Dr. Karimianpour is a Cardiac Electrophysiologist at Piedmont Heart Institute. He is from Virginia where he obtained his medical degree. He completed his residency at Cleveland Clinic, followed by fellowships in Cardiology and Electrophysiology at the Medical University of South Carolina. Dr. Karimianpour has authored several peer-reviewed publications and book chapters. Dr. Karimianpour has a special interest in treatment of ventricular tachycardia and is the director of the Comprehensive VT Center at Piedmont Heart Institute.


