Nearly 38 million people in the United States and 830 million worldwide live with diabetes mellitus (DM), and the burden of this disease in both cost and mortality continues to rise.
Glucose detection is a cornerstone of DM and was initially used to confirm diagnosis before effective anti-hyperglycemic drugs were available. The discovery of hypoglycemia-inducing diabetes treatments such as insulin (1923) and sulfonylureas (1942) presented a new challenge – the need for glucose detection in order to make treatment decisions.
As more drugs to treat DM became available, at-home glucose testing gained popularity and initially involved color-matching urine-reagent-based strips. The first blood glucose strips were developed in 1964 required a large drop of blood (30 μL) and a wait time of nearly 60 seconds.
Blood glucose monitoring (BGM) improved in efficiency and ease of use over the years but remained cumbersome for patients. Insulin users were recommended to check glucose four times a day in order to make treatment decisions safely. Unsurprisingly, patients were often non-compliant as testing was time-consuming, required paraphernalia (strips, lancets, meter) and involved physical discomfort. Failure to test adequately led to risk of overtreatment, resulting in hypoglycemia.
Frequent hypoglycemia can lead to a severe complication called hypoglycemia unawareness. This is a state where one’s ability to feel symptoms of a low glucose at a certain glucose threshold diminishes with recurrent episodes and can result in loss of consciousness, seizure or even death. “Dead in Bed Syndrome” is an unfortunate finding described in insulin users in medical literature and is hypothesized to occur due to nocturnal hypoglycemia triggering fatal cardiac arrhythmias.
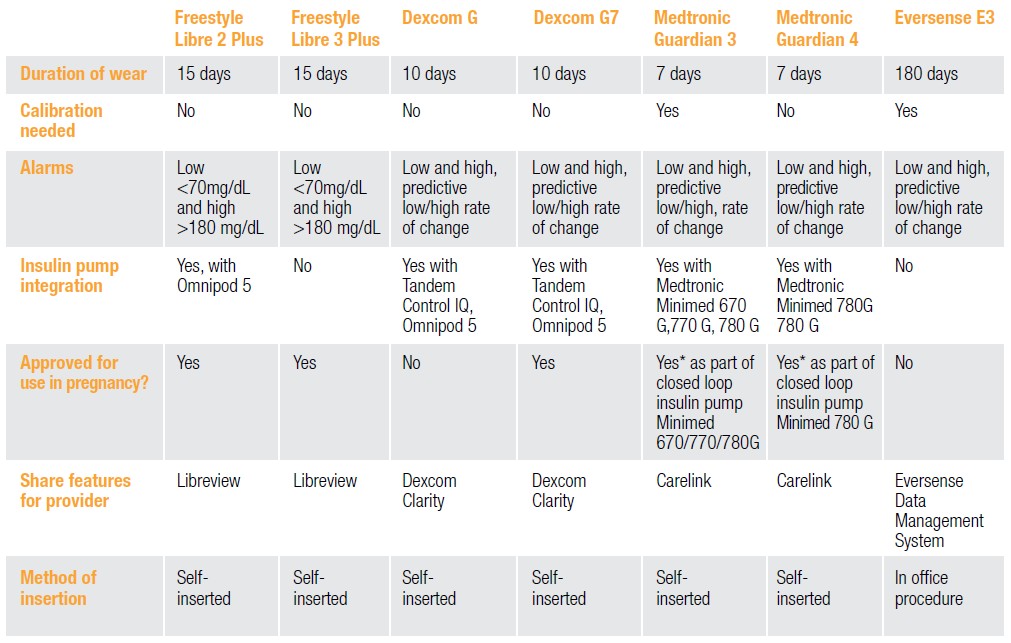
Table 1: Features of the Newest FDA-approved CGMs in the United States
CGMs: Who, What, When?
Continuous Glucose Monitors (CGMs) have been a gamechanger in the field of DM. The first iterations of CGMs were approved by the FDA only as supplementary to BGM and were not to be used for treatment decisions. Medtronic’s Minimed was approved in 1999, followed by Dexcom’s STS in 2006, and Abbott’s Freestyle Navigator in 2008.
The accuracy of CGMs significantly improved over time, leading to FDA approval for its use in treatment decisions and as a replacement for blood glucose testing in 2016.5 The newest FDA approved CGMs include Medtronic’s Guardian Sensor 4, Abbott’s Freestyle Libre 2 plus and 3 plus, and Dexcom’s G6 and G7. All are approved for use in patients with DM over the age of 2, are factory-calibrated and do not need fingerstick confirmation, and can be used to make treatment decisions.
These CGMs are more sophisticated and can generate real- time glucose data. They also have predictive alarms during hypo- and hyperglycemia events and digitally display a glucose trend arrow to help patients better understand their glucose fluctuations.
In multiple studies, CGM users with both Type 1 and Type 2 DM reported an improved quality of life as constant awareness of glucose levels helped them tailor their diet and exercise to achieve goals, improve overall understanding of the disease process and reduce diabetes-related stress. Moreover, the CGM hypoglycemia alarms have proven to be a crucial safety feature in preventing hypoglycemic emergencies. Public confidence in CGMs has improved to the point where the FAA upgraded its guidelines in October 2019 to allow pilots on insulin to apply for first- or second-class mailman certification if using a CGM.
CGM Reports: Time-In-Range is the New Hemoglobin A1c
While CGMs help patients manage their DM better, they can also assist providers in tailoring DM treatment to a patient’s needs. CGM receivers now have the capacity to store months of glucose data and to generate detailed reports which help providers better understand patient compliance and determine gaps in the prescribed treatment.
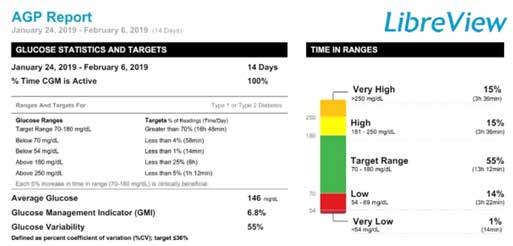
Two important features of these reports that provide detailed data critical to patient care are Time In Range (TIR) and Glucose Management Indicator (GMI). TIR provides more nuanced information than Hemoglobin A1c (HbA1c) as it is an estimation of how much time a patient’s glucose readings stay between 70-180 mg/dL, a range that includes both fasting and postprandial acceptable glucose targets to limit DM complications.
HbA1c is a less-precise average glucose estimation based on glycated hemoglobin over 3 months. Additionally, it fails to identify daily hypo- and hyperglycemic excursions that may contribute to hypoglycemia unawareness and micro- and macrovascular complications.
Due to HbA1c’s reliance on measuring hemoglobin, anemia presents another limitation and is common in patients with chronic conditions and in the elderly. As a nod to HbA1c’s longstanding familiarity in the medical and public realm, CGM reports also provide a helpful HbA1c estimate or Glucose Management Indicator (GMI) value.
CGMs: How Do They Work?
While blood glucose testing relies on glucose measurement in the capillaries, CGMs rely on glucose measurement in the interstitial fluid surrounding the capillaries and cells in our bodies. CGM technology utilizes an enzymatic electrochemical reaction to produce glucose readings and consists of three parts: a sensor, a transmitter and a receiver.
The sensor probe contains glucose oxidase, which catalyzes the oxidation of glucose in the interstitial fluid to hydrogen peroxide (H2O2). Next, an electrical current is produced in proportion to the amount of H2O2 generated and allows the transmitter to convert the signal to a glucose reading displayed in mg/dL on the receiver.
CGM Value vs Blood Glucose Values: Which is More Accurate?
Most patients who transition from blood glucose testing to CGMs will report a “discrepancy” between their blood glucose reading and concurrent CGM reading. This can be a source of confusion and frustration for patients and reduce trust in CGMs.
CGM readings lag behind blood glucose readings by nearly 15-20 minutes because the glucose molecule has to diffuse from the capillary space into the interstitial fluid before being picked up by the CGM sensor.15,16 The values can also be + 15-20 mg/dL due to this lag time, especially when glucose values are rapidly changing.
Confirming CGM values with blood glucose readings can be helpful but is not necessary for most of the newer factory- calibrated CGMs such as Dexcom G6 and G7, Freestyle Libre 2 plus and 3 plus, or Guardian 4. It is recommended to calibrate CGMs when a patient’s symptoms do not match the sensor reading.
Sample LibreView Report:
CGM: Inpatient Use
As the number of hospitalized patients with DM or hyperglycemia has exponentially risen over the past three decades, inpatient glucose control plays a significant factor in healthcare costs. Inpatient hyper- or hypoglycemia is associated with the following:
- Increased risk of post-surgical complications
- Nosocomial infections
- Duration of hospital stay
- Critical care needs
- 30-day readmission rates
- Higher morbidity and mortality
Current inpatient guidelines according to the American Diabetes Association (ADA) include a glucose target of 140-180 mg/dL for most hospitalized patients, treatment with insulin over oral anti-hyperglycemic agents, and glucose monitoring with point-of-care (POC) fingerstick testing at least every 4-6 hours.18 Several studies have shown CGMs are better able to detect asymptomatic or nocturnal hypoglycemia compared to POC glucose testing and is more comfortable for patients.
At present, the FDA recommends continuing using CGMs in hospitalized DM patients already on the device but does not recommend initiating its usage. However, there was an exception to this FDA guideline during the COVID-19 pandemic when CGMs were initiated in both ICU and non-ICU patients to minimize exposure time between patients and healthcare providers. The future of inpatient glucose monitoring may include use of CGMs to remotely track a patient’s glucose similar in concept to telemetry.
Patient example: How to interpret a Dexcom Clarity report
Patient John Doe is on a multiple daily insulin regimen (MDI) and has an estimated HbA1c of 7.3 per the GMI value. The Ambulatory Glucose Profile (AGP) breakdown shows that this patient’s TIR is only 62%, with 36% of glucose values above goal of 180 mg/dL and 2% in the hypoglycemia range below 70 mg/dL. The goal of therapy is to achieve TIR over 85% without any hypoglycemia below 70 mg/dL. The clinician should review proper insulin administration techniques with the patient, including timing prandial insulin ideally 15 minutes before meals to provent post-meal spikes or low glucose values, reduce basal insulin dose if fasting hypoglycemia, reiterate compliance if significant day-to-day variation is noted.
CGM: Access for Patients
Although the ADA and other governing bodies have published studies demonstrating the benefits of CGMs to patients with chronic DM, access has not been easy. A study showed patients with DM who were less likely to have access to CGMs tended to have the following characteristics:
- Older
- Disadvantaged socioeconomically
- Living in the Southeastern United States
- Medicaid recipients
- African American or Hispanic
Moreover, until July 2021 most insurance companies had strict guidelines for CGM coverage – patients needed to be on at least three daily insulin injections and were required to provide a 30-day blood glucose log documenting four daily fingerstick readings.
Thanks to advocacy efforts, CGMs are now approved for most commercially insured patients regardless of insulin use and for all insulin-using recipients of Medicare and Medicaid. Georgia Medicaid was the most recent insurance plan to approve CGMs after Governor Brian Kemp signed legislation expanding access on July 1, 2024.
Despite this progress, the paperwork burden generated from prescribing CGMs remains a limitation for many providers, as most commercial insurances require prior authorization paperwork and Medicare and Medicaid plans require the use of third-party Durable Medical Equipment companies (DME).
The Future of CGMs: From Treatment to Prevention
The FDA approved STELO from Dexcom in March 2024 as the first-ever CGM intended for use in those with or without DM. This CGM provides real-time glucose data without any hyper- or hypoglycemia alarms, therefore is not intended for patients with DM on hypoglycemic drugs or those with a history of hypoglycemia.
For those with obesity, pre-diabetes or a family history of diabetes that want to know how food affects their glucose levels, STELO can be used as a preventative tool and can be purchased without a prescription. Having this knowledge can lead to dietary adjustments such as reducing consumption of processed and ultra-processed foods and incorporating healthy fats, whole grains and legumes with higher fiber content to prevent post-meal glucose spikes.
 Dr. Rahman is a board-certified endocrinologist with Piedmont Healthcare, providing comprehensive care to patients in Rockdale and the surrounding counties. She earned her MD at LSU Health Sciences in New Orleans, followed by an internal medicine residency at University of Arkansas and an endocrinology fellowship at the University of Maryland. She is part of Piedmont’s Diabetes Taskforce and believes in a patient-centered approach, with a special interest in improving access for low-income populations with diabetes and obesity.
Dr. Rahman is a board-certified endocrinologist with Piedmont Healthcare, providing comprehensive care to patients in Rockdale and the surrounding counties. She earned her MD at LSU Health Sciences in New Orleans, followed by an internal medicine residency at University of Arkansas and an endocrinology fellowship at the University of Maryland. She is part of Piedmont’s Diabetes Taskforce and believes in a patient-centered approach, with a special interest in improving access for low-income populations with diabetes and obesity.