It’s been over three decades since genetic testing was first introduced as a tool for patients at risk of hereditary cancer syndromes. In its early days, genetic testing was expensive, time-consuming (often taking up to eight weeks for results) and offered limited impact on clinical management. Fast forward to 2026, genetic testing is now a routine part of cancer care. It’s more accessible, comprehensive and, perhaps most importantly, significantly impactful for patients and their families.
Midlife: it is a powerful biological transition that intersects with cardiovascular health, brain health, bone health, metabolic health, and overall longevity. It represents a pivotal biological and clinical inflection point for women.
For many, the menopause transition coincides with the acceleration—or new onset—of chronic disease. For women of color, particularly Black women, this intersection is not incidental. It reflects a convergence of biology, social determinants of health, structural inequities, and cumulative stress exposure that shapes both symptom burden and long-term health outcomes.
As physicians, we must recognize that menopause does not occur in isolation. It unfolds against the backdrop of cardiometabolic risk, autoimmune disease, mental health disorders, and persistent disparities in access, diagnosis, and treatment. Caring well for women of color in midlife requires integrating menopause management with chronic disease care—through an explicitly equity-focused lens. Menopause—particularly for women of color—represents one of the next frontiers of health equity in America.
 Menopause Is Not a Uniform Experience
Menopause Is Not a Uniform Experience
The Study of Women’s Health Across the Nation (SWAN), a multiethnic longitudinal cohort, has provided foundational insight into racial differences in menopausal experience. African American women experience vasomotor symptoms (VMS) more frequently and with greater severity, endure them for a median of 10.1 years—nearly twice as long as White women—and enter menopause earlier than several other racial groups.
These disparities are not fully explained by biology. SWAN data document differential exposure to adverse life events, financial strain, discrimination, and structural stressors—the cumulative physiologic toll of which drives the weathering process, accelerating biological aging and elevating allostatic load in ways that amplify symptom burden before, during, and after the menopausal transition.¹ A narrative review of African American women’s menopausal experiences further highlights distinct differences in physical, psychological, and quality-of-life domains that extend well beyond VMS frequency and duration alone.² Importantly, while African American women report higher VMS prevalence in cohort studies, commercial claims data reveal lower rates of diagnosed VMS among Black, Hispanic, and Asian women compared to non-Hispanic White women—a discrepancy that strongly indicates underdiagnosis and unmet clinical need.³
The Chronic Disease Burden in Midlife
Midlife is also when cardiometabolic risk accelerates. Women of color disproportionately experience hypertension, type 2 diabetes, obesity, chronic kidney disease, autoimmune conditions, depression, and anxiety.4
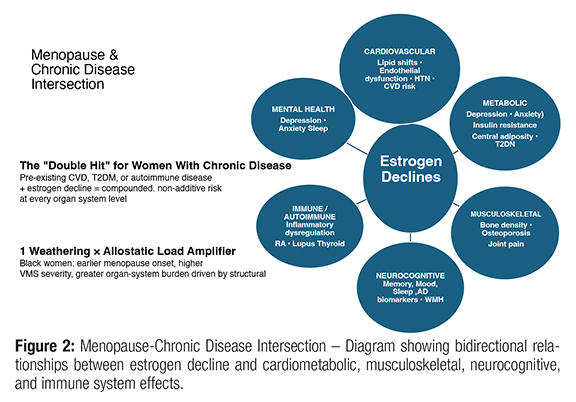
Estrogen plays a regulatory role across multiple organ systems—cardiovascular, metabolic, musculoskeletal, neurocognitive, and immune. Its decline contributes to adverse lipid shifts, endothelial dysfunction, central adiposity, and bone loss.9 For women already managing chronic disease, menopause can represent a “double hit”: worsened glycemic control, exacerbated hypertension, accelerated bone loss in patients on chronic steroids, intensified autoimmune symptoms, and greater cardiovascular risk in those with existing metabolic syndrome (Fig 2).
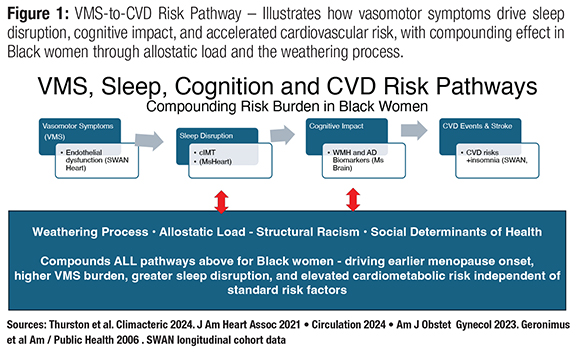
 Vasomotor Symptoms and Cardiovascular Risk
Vasomotor Symptoms and Cardiovascular Risk
VMS are no longer regarded as incidental quality-of-life symptoms. A growing body of evidence—anchored by SWAN and the MsHeart/MsBrain studies—has established frequent or persistent VMS as independent markers of adverse cardiovascular health. In SWAN Heart, women reporting hot flashes demonstrated poorer endothelial function and greater aortic calcification independent of standard CVD risk factors. In MsHeart, more frequent objectively-assessed VMS were associated with higher carotid intima-media thickness (cIMT) and greater carotid plaque in a dose-response relationship. Critically, the variance in cIMT attributable to VMS frequency exceeded that of any single standard CVD risk factor. The SWAN cohort further demonstrated that persistent VMS are associated with increased risk for incident CVD events, including myocardial infarction and stroke.5
Sleep disruption compounds this risk substantially. VMS—especially nocturnal hot flashes—are strongly and consistently associated with all dimensions of sleep disturbance in SWAN analyses. In MsHeart, shorter objective sleep duration was independently associated with greater carotid atherosclerosis even after adjusting for CVD risk factors, hot flashes, and estradiol. A landmark SWAN analysis tracking sleep trajectories over 22 years found that chronic insomnia symptoms across midlife significantly elevated CVD event risk later in life—with the greatest hazard among women who also had chronically short sleep duration.6 Sleep disruption also carries cognitive consequences: objectively measured nocturnal VMS have been linked to white matter hyperintensities and adverse Alzheimer’s disease biomarkers—including lower plasma Aß42/Aß40 ratios—in the MsBrain study, identifying VMS as a potentially modifiable risk factor for cognitive decline.7
For Black women, who bear disproportionate VMS burden and the compounding weight of weathering-related allostatic load, these intersecting pathways—VMS, sleep disruption, cognitive vulnerability, and CVD risk—warrant urgent clinical attention. (Fig 1) Yet claims data reveal that Black, Hispanic, and Asian women are more likely to discontinue VMS treatment than White women, and lower income is independently associated with higher discontinuation rates.³ Structural barriers, therapeutic mistrust, and inadequate shared decision-making drive a pattern where the women most at risk are the least likely to sustain treatment.3
 Underdiagnosis and Clinical Silence
Underdiagnosis and Clinical Silence
The gap between symptom burden and documented diagnosis is a critical failure point. In commercial claims data (2017–2020), the 4-year prevalence of diagnosed VMS was 82.5 per 1,000 among non-Hispanic White women versus 71.6 per 1,000 among Black women—despite SWAN data showing Black women report higher VMS frequency and severity.³
This disconnect reflects a constellation of barriers: less frequent help-seeking, shorter clinical visits, provider under recognition, cultural framing of menopause as “natural” rather than medical, and menopause displaced by competing priorities during chronic disease encounters. Survey data confirm that many women feel rushed or unheard during clinical visits.8 Provider attitudes, implicit bias, and insufficient training in menopause medicine each contribute to the silence. If the menopause discussion never occurs, the diagnosis is never coded—and the patient’s suffering remains invisible.
Clinical Complexity: Distinguishing Symptoms
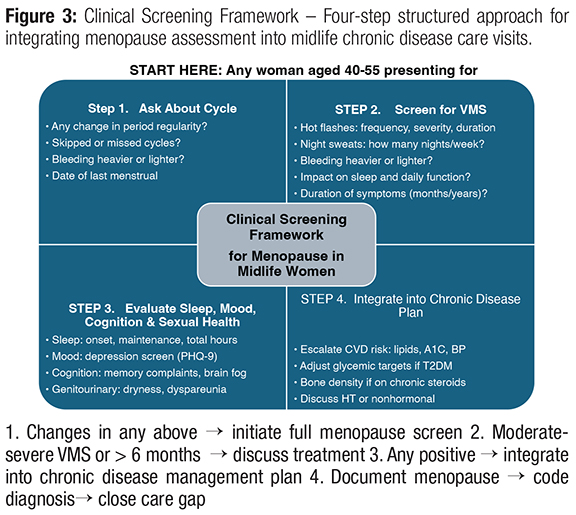
For women with chronic illness, differentiating menopause-related symptoms from disease activity is challenging. Hot flashes may be mistaken for anxiety; sleep disruption attributed solely to depression; cognitive changes labeled as stress; palpitations referred to cardiology without considering hormonal transition. Physicians must actively screen midlife women—especially those aged 40–55 with diabetes, autoimmune disease, or cardiovascular risk factors. A structured clinical approach (Fig 3):
1. Ask directly about menstrual cycle changes.
2. Screen for VMS severity and duration.
3. Evaluate sleep, mood, cognition, and sexual health.
4. Integrate findings into chronic disease management plans.
Treatment Options and Equity
Hormone therapy (HT) remains the most effective treatment for moderate-to-severe VMS and, for appropriately selected patients within 10 years of menopause onset, may also favorably affect lipid profiles and vascular function. Transdermal formulations carry a lower thrombotic risk profile and may be preferred in women with hypertension or obesity. Yet women of color are less likely to receive or continue HT, due to historical mistrust, residual WHI concerns, cost, limited counseling time, and provider discomfort.
Nonhormonal options—including SSRIs, SNRIs, gabapentin, and the newer NK3 receptor antagonist fezolinetant and NK1 & 3 receptor antagonist elinzanetant—offer additional pathways. The principle is clear: menopause treatment is not elective comfort care. It is chronic disease mitigation and quality-of-life preservation.
 Allostatic Load and the Weathering Framework
Allostatic Load and the Weathering Framework
The weathering hypothesis posits that chronic exposure to racial stressors accelerates biological aging through allostatic load—the cumulative physiologic burden sustained across cardiovascular, metabolic, and immune systems.¹ SWAN data confirm that African American women accumulate higher allostatic load scores across midlife than White women, independent of poverty. This framework contextualizes earlier and more symptomatic menopausal transitions, elevated inflammatory markers, and greater cardiometabolic vulnerability.9 Screening for social determinants of health and stress burden must be central—not peripheral—to midlife care.
Digital Health and Access Gaps
Survey data show that many midlife women seek menopause information online, yet nearly half report dissatisfaction with available digital resources.8 For communities facing transportation barriers or specialist shortages, digital platforms may improve education, support symptom tracking, and facilitate telehealth follow-up. Digital literacy disparities and broadband inequities must be addressed to avoid widening existing gaps.
What Atlanta Physicians Can Do Now
Metro Atlanta serves one of the most diverse populations in the United States. The following six actions can meaningfully improve care for women of color in midlife:
1. Normalize the conversation. Proactively ask about menopause beginning at age 40. Do not wait for patients to raise the issue.
2. Integrate care. Embed menopause screening into chronic disease visits—especially for hypertension, diabetes, and autoimmune disorders.
3. Address treatment equity. Discuss both hormonal and nonhormonal options; explore concerns directly; review contraindications thoughtfully rather than reflexively.
4. Monitor cardiometabolic risk. Use menopause as an opportunity to assess lipids, A1C, blood pressure, and physical activity.
5. Screen for sleep and mood. Persistent VMS often coexists with sleep disturbance and depression—both of which worsen chronic disease outcomes.
6. Build trust. Allocate time. Listen. Validate symptoms. Cultural humility and relational continuity are evidence-based practices.
The Imperative for Action
Menopause affects approximately 6,000 women daily in the United States. Black women and other women of color often experience longer symptom duration, higher chronic disease burden, and lower treatment persistence. Failing to recognize and address menopause in this population compounds existing inequities in cardiovascular disease, osteoporosis, mental health, and quality of life.
Caring for women of color in midlife is about preserving cardiovascular health, protecting bone and brain function, sustaining workforce participation, honoring lived experience, and advancing health equity. Menopause is a window of opportunity. For women of color living with chronic disease, it is also a window of vulnerability. Our responsibility—as Atlanta physicians and as a medical community—is to ensure it becomes a window for intervention, not neglect.
References
1. Geronimus AT, Hicken M, Keene D, Bound J. “Weathering” and age patterns of allostatic load scores among Blacks and Whites in the United States. Am J Public Health. 2006;96(5):826–833. doi:10.2105/AJPH.2004.060749
2. Williams M, Richard-Davis G, Weickert V, Christensen L, Ward E, Schrager S. A review of African American women’s experiences in menopause. Menopause. 2022;29(11):1331–1337. doi:10.1097/GME.0000000000002060
3. Richard-Davis G, Singer A, King DD, Mattle L. Understanding attitudes, beliefs, and behaviors surrounding menopause transition: results from three surveys. Patient Relat Outcome Meas. 2022;13:271–283. doi:10.2147/PROM.S375144
4. Marlatt KL, Redman LM, Beyl RA, et al. Racial differences in body composition and cardiometabolic risk during the menopause transition: a prospective, observational cohort study. Am J Obstet Gynecol. 2020;222:365.e1–18.
5. Thurston RC, Karvonen-Gutierrez CA, Derby CA, et al. Vasomotor symptoms and cardiovascular health: findings from the SWAN and the MsHeart/MsBrain studies. Climacteric. 2024;27(1):75–80.
6. Thurston RC, Guo M, Chang Y, et al. Trajectories of sleep over midlife and incident cardiovascular disease events in SWAN. Circulation. 2024;149(7):545–555. doi:10.1161/CIRCULATIONAHA.123.064431
7. Thurston RC, Aizenstein HJ, Derby CA, et al. Menopausal vasomotor symptoms and plasma Alzheimer disease biomarkers. Am J Obstet Gynecol. 2023. doi:10.1016/j.ajog.2023.08.049
8. Richard-Davis G, Singer A, King DD, Mattle L. Understanding attitudes, beliefs, and behaviors surrounding menopause transition: results from three surveys. Patient Relat Outcome Meas. 2022. doi:10.2147/PROM.S375144
9. Thurston RC, El Khoudary SR, Sutton-Tyrrell K, et al. Menopausal vasomotor symptoms and risk of incident cardiovascular disease events in SWAN. J Am Heart Assoc. 2021;10(3):e017416.


