Over the past decade there’s been a massive increase in the development of diabetes medications. The new approach in prescribing type 2 diabetes medications is to consider all aspects of the patient’s health, such as their cardiovascular event risk, kidney health and other comorbidities.
The following are the highlights from AACE guidelines on the complications-centric algorithm for glycemic control. (See Figure 1, page 14.)
- Individualize Glycemic Target (A1c <6.5% for most or 7%-8% if high risk or adverse consequences from hypoglycemia and/or limited life expectancy)
- HbA1c >7.5 % start 2 agents, for A1c >9.0% or >1.5% above goal start 2-3 agents
- HbA1c >10% and/or glucose >300mg/dl with symptoms then use basal insulin +/- GLP-1 RA
The first diabetic medication to start is metformin unless there is a contraindication such as a low glomerular filtration rate (GFR), GI side effects or adverse reactions.
It is best to slowly up titrate metformin, starting at 500 mg daily, then increase by 500mg every few days until reaching the target dose of 1000 mg twice per day. If the patient develops diarrhea or abdominal discomfort, they can try metformin ER 500 mg tablets. The patient will decide the highest tolerated dose.
Most insurance plans will deny glucagon-like peptide-1 receptor agonists (GLP-1RA) and other high-cost diabetes medications if the patient does not have type 2 diabetes and there is no documentation of use, or contraindications to, metformin.
In patients with chronic kidney disease (CKD), the following doses can be used: for a GFR of 30-45 metformin 500 mg daily, for a GFR of 45-59 metformin 500 mg bid, and for a GFR >60 metformin 1000 mg bid.
The second or third diabetes medication choice depends on the patient’s comorbidities. For example, for atherosclerotic cardiovascular disease (ASCVD) or high risk of cardiovascular disease (CVD), select a GLP-1 RA or SGLT2 inhibtor. For CKD, select a GLP-1 RA or SGLT2 inhibtor. For stroke/TIA, select a GLP-1 RA or pioglitazone, and for heart failure select a SGLT2 inhibitor.
What are GLP-1 and GIP?
The primary role of GLP-1 and gastric inhibitory polypeptide (GIP) is glucose reduction. They are naturally occurring hormones that are synthesized in the small intestines and result in a glucose-dependent stimulation of insulin secretion. With GLP-1 there is a reduction in gastric emptying, which helps contribute to satiety.
The mechanism of action of GLP-1-based therapies (GLP-1 receptor agonist, dual-actin GLP-1 and GIP receptor agonist, DPP4 Inhibitor) is that they work on receptors where GLP and GIP would normally act and work to enhance glucose-dependent insulin secretion, slowed gastric emptying and reduction of post postprandial glucagon. As result, in patients who take these therapies, there is a great reduction in postprandial hyperglycemia.
How to Choose the Correct GLP-1/GIP Medication
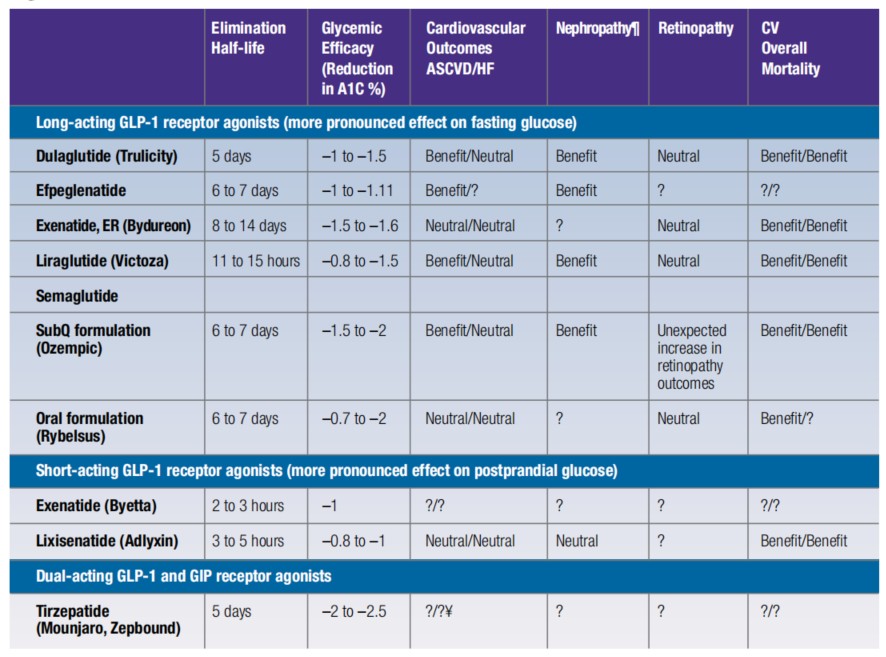
Of all the GLP-1 RAs, dulaglutide (Trulicity), liraglutide (Victoza) and semaglutide SQ (Ozempic) have shown benefit in CV outcomes, CV overall mortality and nephropathy benefits. What are the names and what are the differences? (See Figure 1, page 14.)
- Basal insulin combination (prespecified ratio)
- Insulin degludec and liraglutide (Zultophy= Tressiba + Victoza)
- Insulin glargine and lixisenatide (Soliqua = Lantus + Adlyxin)
Figure 1. GLP-1 RAs
Long-acting GLP-1 receptor agonists Dulaglutide (Trulicity)
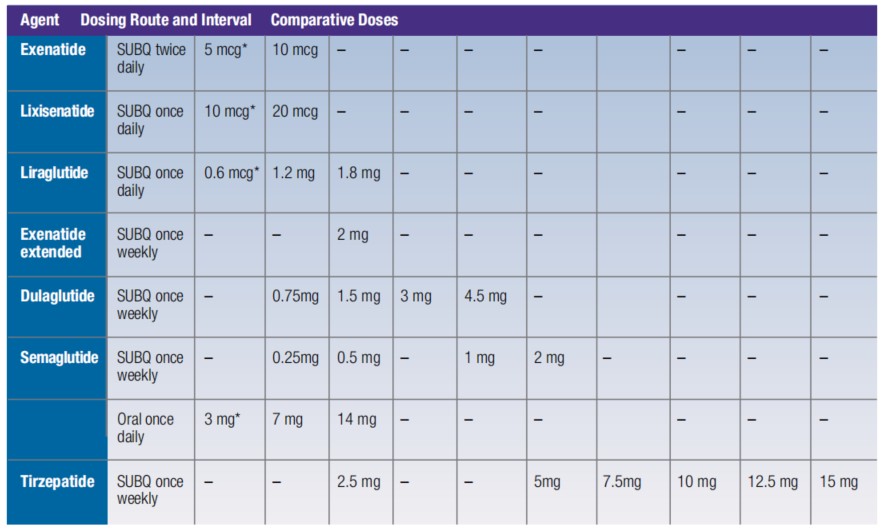
Initial dose: 0.75 mg SubQ once weekly. If not meeting glycemic goals after 4 weeks, increase dose as tolerated every 4 weeks as follows: 1.5 mg SubQ once weekly, then 3 mg SubQ once weekly, then 4.5 mg SubQ once weekly.
Exenatide, extended release (Bydureon)
Initial dose: 2 mg SubQ once weekly.
Liraglutide
Initial dose: 0.6 mg SubQ once daily for 1 week, then increase to 1.2 mg SubQ once daily. If not meeting glycemic goals after another 1 to 2 weeks, increase dose as tolerated to 1.8 mg SubQ once daily.
Semaglutide SubQ formulation (Ozempic)
Initial dose: 0.25 mg SubQ once weekly for 4 weeks, then increase to 0.5 mg SubQ once weekly; if not meeting glycemic goals after at least 4 weeks, increase dose as tolerated every 4 weeks as follows: 1 mg SubQ once weekly, then 2 mg SubQ once weekly.
Semaglutide Oral formulation (Rybelsus)
Initial dose: 3 mg once daily for 30 days, then increase to 7 mg once daily. If not meeting glycemic goals after 30 days on 7 mg dose, increase dose as tolerated to 14 mg once daily.
Short-acting GLP-1 receptor agonists Exenatide(Byetta)
Initial dose: 5 mcg SubQ twice daily
If not meeting glycemic goals after 4 weeks, increase dose as tolerated to 10 mcg SubQ twice daily.
Lixisenatide (Adlyxin)
Initial dose: 10 mcg SubQ once daily, within 1 hour prior to any meal of the day.
After 2 weeks, increase dose as tolerated to 20 mcg SubQ once daily
Tirzepatide (Mounjaro)
Initial dose: 2.5 mg SubQ once weekly for 4 weeks, then increase to 5 mg SubQ once weekly. If not meeting glycemic goals after at least 4 weeks, increase dose as tolerated in 2.5 mg increments every 4 weeks as needed to achieve glycemic goals. Maximum dose 15 mg SubQ once weekly.
With the advent of many new diabetes medications, the management of type 2 diabetes has changed drastically over the past decade. These new therapies overall have not only improved diabetes control, but have also help reduce weight, cardiovascular risk and kidney health.
Figure 2. GLP-1 RA-based Drug Shortages and Suggested Comparative Doses for Treating Type 2 Diabetes
 Dr. Patel received her medical degree from Wayne State University School of Medicine. She completed her Internal Medicine Residency and her Endocrinology Fellowship at Case Western Reserve University School of Medicine. She practices medicine at Piedmont Endocrinology and is board certified by the American Board of Internal Medicine in Endocrinology, Diabetes and Metabolism. Dr. Patel specializes in management of adults with disorders of the endocrine system.
Dr. Patel received her medical degree from Wayne State University School of Medicine. She completed her Internal Medicine Residency and her Endocrinology Fellowship at Case Western Reserve University School of Medicine. She practices medicine at Piedmont Endocrinology and is board certified by the American Board of Internal Medicine in Endocrinology, Diabetes and Metabolism. Dr. Patel specializes in management of adults with disorders of the endocrine system.