
A number of factors contribute to the lack of Black patients’ participation in clinical trials, including the mistrust of healthcare systems, lack of access to medical care, low health literacy and lack of awareness about research opportunities.
The enrollment in clinical trials among disadvantaged minority groups in the United States has remained low despite the National Institutes of Health (NIH) Revitalization Act of 1993, which aimed to increase such enrollment in NIH-sponsored clinical trials.1,2 Less than 2% of NIH-sponsored trials are focused on racial and ethnic minority groups.3 More specifically, Black people make up less than 5% of participants in clinical trials4 even though they comprise about 13% of the United States population.5
Table 1.
Patient Sociodemographic Characteristics (N= 40).
| Sociodemographic Characteristics of Sample | Percentages |
| Gender
Women Men |
77.5% 22.5% |
| Education Level
Bachelor’s degree and above Less than a Bachelor’s degree |
60% 40% |
| Average Age (Years)
Age Range |
42
20-76 |
| Median Household Income (per year) | $60,000 |
| Marital Status
Married Divorced Never Married Living with Partner Separated Widowed Other |
35% 15% 37.5% 5% 2.5% 2.5% 2.5% |
| Employed
Yes No Not Applicable |
70% 28% 2% |
| Have Children
Yes No |
60% 40% |
Low participation in clinical trials is believed to primarily stem from historic roots. African Americans’ mistrust of the medical system is believed to play a significant role in trial participation.6
The most well-known study considered to be an abuse of patients’ trust was the Tuskegee Syphilis Study.7 In this study, African American men with syphilis, who were the participants in the study, did not receive any treatment for their syphilis even when a cure was known, suffering long-term harm. Due to such historical abuse by the medical community, mistrust among minority groups, but particularly among African Americans, are believed to be major barriers to participation in clinical research.8
Trust is widely recognized as a central feature in effective patient-physician interaction and intimately linked to a successful patient-physician relationship.9-12 Fundamentally, trust is the central driver of meaning, importance and substance in the patient-physician relationship.13 Patient trust has been linked to a variety of key behaviors and attitudes such as willingness to seek care, adherence to treatment regimes and participation in clinical trials.14-16

While research on patient trust, race and social class has increased over the last three decades, investigations on the degree to which African-American inflammatory bowel disease (IBD) patients trust their doctors is extremely limited. Of course, there are other factors affecting the participation of Black people in medical research including social factors such as logistical issues, health literacy and lack of access to good medical care. 8,17,18
IBD is a chronic recurrent inflammatory condition that can affect the entire gastrointestinal tract.19 Research on the care of IBD patients has been primarily in populations of European ancestry. However, recent studies indicate a potential shift in the epidemiology of IBD in the U.S., with significant increases in the incidence rates among non-White patients.20,21 Despite the dramatic increases in the incidence of IBD among minority groups, their representation in IBD clinical trials and research studies is limited. 22,23
A consensus conference on the care of Black and Hispanic IBD patients was organized by the Morehouse School of Medicine in 2021. Scholars at the conference concluded that there were significant gaps in knowledge about these patient populations.24
Data are sparse on medical, endoscopic and surgical outcomes, as well as the rapid increase in the incidence of IBD among these patient groups. So we conducted a survey of Black IBD patients who were participants of the 2021 Equity in Inflammatory Bowel Disease conference, hosted by Color of Crohn’s and Chronic Illness (COCCI), to identify potential barriers to research, such as mistrust of providers, healthcare systems, and medical research. The conference participants included patient members who registered for the event and attended either in person or virtually. The resulting survey data sheds light on the patients’ lives and, more specifically, on their interactions with the medical community.
Data and Methods
The patient survey was developed to evaluate patients’ trust in their physicians, their experiences with medical and healthcare systems, and their involvement with clinical trials. The survey instrument was constructed by modifying the Corbie-Smith Distrust in Clinical Research Index25 and the Trust in Physician Scale.9 The survey was reviewed and approved by the Institutional Review Board of Morehouse School of Medicine.
Links to the survey were sent out via SurveyMonkey to the participants of the patient education conference. The conference was held in a hybrid format, so participants were both in person and online. Patient participants, who were confirmed members of COCCI, were contacted via email and text message.
Survey correspondence was sent to the attendees before, during and one week after the education conference. We used descriptive statistics to describe the main characteristics of the patients and cross-tabulations to probe a number of issues.
Survey Results
In all, the survey response rate was 64% (48 out of 75 patients), of which 83% respondents identified as Black or African American; we focused our analysis specifically on the latter group (n=40).

Table 1 provides details of the sample of African Americans selected from the larger sample. Of note, among these 40 respondents registered for the conference, about 78% identified as a woman with a median age of 42, 60% of the sample had earned a bachelor’s degree with an overall median income of $60,000. 35% of the sample was married with another 38% never married. 70% were employed, and 60% had at least one child.
The survey findings show high levels of trust of healthcare providers, which contradicts the documented perceptions of mistrust by Black patients.8 Over 80% of this sample of Black patients trusted their primary care physician and their gastroenterologist (see Figure 1), which suggests a strong sense of trust among these respondents.
One of the most frequently cited barriers to clinical trial participation among Black people is the lack of trust in the healthcare system and biomedical research, which is attributed to historical abuses such as the Tuskegee Syphilis Study.7 The “legacy of the Tuskegee Syphilis Study” is a phrase in the literature that indicates Black patients are more reluctant to participate in biomedical research studies because of the infamous study.6,26
However, among Black IBD patients in our survey, the study does not play a substantial role in their participation. Similar to previous reports26, 85% of our patient respondents have heard of the Tuskegee study. However only 28% of them indicated that it would play a role in their decision to participate in a clinical trial.
Still, although 46% of the sample were invited to participate in clinical research, only 18% did so. (See Figure 3.) The lower rate of research participation may be due, at least in part, to the duration of the patients under the care of gastroenterologists. (See Figure 4.) The longer a patient had been with their gastroenterologist, the more likely they had been invited to participate in medical research.
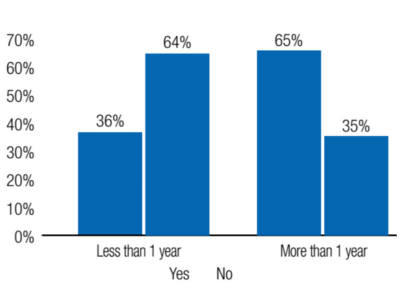
Length of Treatment by Gastroenterologist (n=40).
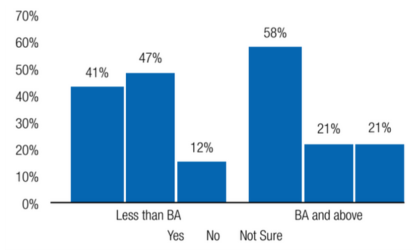
We did find differences by social class in that patients with less than a college degree report less discrimination and racism in the medical field (41%) than those with a college degree (58%). (See Figure 2.) These results indicate that there are different perceptions about racism across social class groupings of African Americans, which suggests that the answer regarding Blacks’ participation in medical research may have to do less with overall perceptions and more with not treating the community as a monolith and digging deeper into the complexity of the Black population in the United States.
The strength of this study is that we asked in-depth questions from a mostly African-American sample and focused our analysis on those who identified as Black or African American. The main limitation of our study is that it is a small, non-random sample collected among individuals attending an education who are likely quite invested in learning more about their disease. Nonetheless, our results indicate a high level of trust for physicians among these Black IBD patients.
In the end, only by ensuring adequate representation of Black patients in clinical research can we reach health equity across the healthcare continuum. Improved access to clinical trials may result in increased representation of Black and other under-represented patient populations in research, which will continue to move the field of IBD forward.
References
- Chen, M.S., Jr., Lara, P.N., Dang, J.H., Paterniti, D.A. & Kelly, K. Twenty years post-NIH Revitalization Act: enhancing minority participation in clinical trials (EMPaCT): laying the groundwork for improving minority clinical trial accrual: renewing the case for enhancing minority participation in cancer clinical trials. Cancer 120 Suppl 7, 1091-1096 (2014).
- Knepper, T.C. & McLeod, H.L. When will clinical trials finally reflect diversity? Nature 557, 157-159 (2018).
- Al Hadidi, S., Mims, M., Miller-Chism, C.N. & Kamble, R. Participation of African American Persons in Clinical Trials Supporting U.S. Food and Drug Administration Approval of Cancer Drugs. Ann Intern Med 173, 320-322 (2020).
- FDA Drug Trials Snapshots Summary Report: Five-Year Summary and Analysis of Clinical Trial Participation and Demographics (2020).
- Census, U.S. 2022. https://www.census.gov/quickfacts/fact/table/US/PST045219.
- Katz, R.V. et al. The Tuskegee Legacy Project: willingness of minorities to participate in biomedical research. J Health Care Poor Underserved 17, 698-715 (2006).
- J.H., J. Bad blood: the Tuskegee Syphilis Experiment. . New York: Free Press (1981).
- Hunninghake, D.B., Darby, C.A. & Probstfield, J.L. Recruitment experience in clinical trials: literature summary and annotated bibliography. Control Clin Trials 8, 6S-30S (1987).
- Anderson, L.A. & Dedrick, R.F. Development of the Trust in Physician scale: a measure to assess interpersonal trust in patient-physician relationships. Psychol Rep 67, 1091-1100 (1990).
- Becker, E.R. & Roblin, D.W. Translating primary care practice climate into patient activation: the role of patient trust in physician. Med Care 46, 795-805 (2008).
- Dugan, E., Trachtenberg, F. & Hall, M.A. Development of abbreviated measures to assess patient trust in a physician, a health insurer, and the medical profession. BMC Health Serv Res 5, 64 (2005).
- Rowe, R. & Calnan, M. Trust relations in health care–the new agenda. Eur J Public Health 16, 4-6 (2006).
- Hall, M.A., Dugan, E., Zheng, B. & Mishra, A.K. Trust in physicians and medical institutions: what is it, can it be measured, and does it matter? Milbank Q 79, 613-639, v (2001).
- Rhodes, R. & Strain, J.J. Trust and transforming medical institutions. Camb Q Healthc Ethics 9, 205-217 (2000).
- Safran, D.G. et al. Linking primary care performance to outcomes of care. J Fam Pract 47, 213-220 (1998).
- Shavers, V.L., Lynch, C.F. & Burmeister, L.F. Racial differences in factors that influence the willingness to participate in medical research studies. Ann Epidemiol 12, 248-256 (2002).
- Freimuth, V.S. et al. African Americans’ views on research and the Tuskegee Syphilis Study. Soc Sci Med 52, 797-808 (2001).
- Owens, O.L., Jackson, D.D., Thomas, T.L., Friedman, D.B. & Hebert, J.R. African American men’s and women’s perceptions of clinical trials research: focusing on prostate cancer among a high-risk population in the South. J Health Care Poor Underserved 24, 1784-1800 (2013).
- Cho, J.H. & Abraham, C. Inflammatory bowel disease genetics: Nod2. Annu Rev Med 58, 401-416 (2007).
- Xu, F., Carlson, S.A., Liu, Y. & Greenlund, K.J. Prevalence of Inflammatory Bowel Disease Among Medicare Fee-For-Service Beneficiaries – United States, 2001-2018. MMWR Morb Mortal Wkly Rep 70, 698-701 (2021).
- Aniwan, S., Harmsen, W.S., Tremaine, W.J. & Loftus, E.V., Jr. Incidence of inflammatory bowel disease by race and ethnicity in a population-based inception cohort from 1970 through 2010. Therap Adv Gastroenterol 12, 1756284819827692 (2019).
- Cohen, N.A., Silfen, A. & Rubin, D.T. Inclusion of Under-represented Racial and Ethnic Minorities in Randomized Clinical Trials for Inflammatory Bowel Disease. Gastroenterology 162, 17-21 (2022).
- Sedano, R. et al. Underrepresentation of Minorities and Lack of Race Reporting in Ulcerative Colitis Drug Development Clinical Trials. Inflamm Bowel Dis 28, 1293-1295 (2022).
- Liu, J.J. et al. The Current State of Care for Black and Hispanic Inflammatory Bowel Disease Patients. Inflamm Bowel Dis 29, 297-307 (2023).
- Corbie-Smith, G., Thomas, S.B. & St George, D.M. Distrust, race, and research. Arch Intern Med 162, 2458-2463 (2002).
- Katz, R.V. et al. The legacy of the Tuskegee Syphilis Study: assessing its impact on willingness to participate in biomedical studies. J Health Care Poor Underserved 19, 1168-1180 (2008).


